What is Erythremia?
Erythremia is a chronic leukemia with a lesion at the level of the cell – the precursor of myelopoiesis with the tumor’s unlimited growth of this cell, which has retained the ability to differentiate between 4 sprouts, mainly red. At certain stages of the disease, and sometimes from the very beginning, myeloid metaplasia in the spleen joins the proliferation of cells in the bone marrow.
Pathogenesis during Erythremia
No specific cytogenetic abnormalities with erythremia were found.
Quantitative chromosome defects, structural aberrations are clonal in nature and are not found in lymphocytes. In patients treated with cytostatics, they are more common. According to the authors, patients with initially
detected violations of the chromosome set are not predisposed to a more malignant course of the disease.
Although there are no morphological, enzymatic, and cytogenetic signs of damage to the lymphatic system in erythremia, the functional state of T-lymphocytes has been changed: a reduced response to known mitogens and an increase in their spontaneous activity have been detected.
In the erythremic stage in the bone marrow, a complete violation of the structure of the sprouts with the replacement of fat is usually observed.
In addition to this classic variant, 3 more types of changes can be observed: an increase in erythroid and megakaryocytic germs, an increase in erythroid and granulocyte germs; an increase in predominantly erythroid germ. Stocks of iron in the bone marrow are significantly reduced. The hematopoietic bridgehead is often expanded, the adipose bone marrow may look red, hematopoietic.
The spleen is overflowing with blood, contains sections of heart attacks of various prescriptions, platelet aggregates, and often initial, moderate or significant signs of myeloid metaplasia with localization in the sinuses. The follicular structure is usually preserved.
In the liver, along with plethora, there are foci of fibrosis, the connection of hepatic beams, sometimes myeloid metaplasia with localization in sinusoids. Very thick bile and pigmented stones are often visible in the gallbladder.
A frequent find are urate stones, pyelonephritis, shriveled kidneys, a significant pathology of their vessels.
In the anemic stage of the disease, there is a pronounced myeloid transformation of the spleen and liver, as well as their increase. The bone marrow is often fibrosed. At the same time, myeloid tissue can be both hyperplastic and reduced, bone marrow vessels are sharply increased in number and structurally altered. Dystrophic and sclerotic changes are detected in parenchymal organs. There are frequent manifestations of thrombotic syndrome or hemorrhagic diathesis.
The functional state of erythrocyte production, according to radiological studies, is sharply enhanced: the half-life of radioactive iron introduced into the vein is shortened, its utilization by the bone marrow is strengthened, and the circulation is accelerated.
The average lifespan of platelets is often shortened, there is a negative relationship between their survival and the size of the spleen.
Symptoms of Erythremia
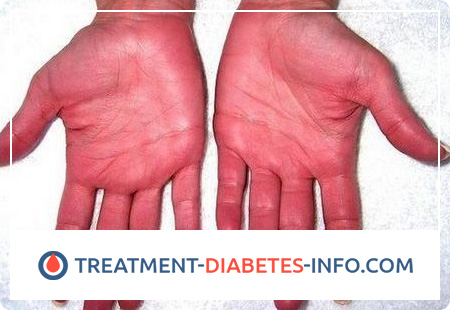
The disease begins gradually. Redness of the skin, weakness, heaviness in the head, enlargement of the spleen, arterial hypertension increase, and in half of patients – excruciating skin itching after washing, washing, swimming. Sometimes the first manifestations of the disease are finger necrosis, thrombosis of the larger arteries of the lower and upper extremities, thrombophlebitis, thrombotic stroke, myocardial or pulmonary infarction, and especially acute burning pains in the fingertips, eliminated by acetylsalicylic acid for 1-3 days. Many patients, long before the diagnosis was established, had bleeding after tooth extraction, skin itching after bathing and “good” red blood counts, which the doctors did not attach due importance to.
In stage I, the duration of which is 5 years or more, a moderate increase in circulating blood is observed, the spleen is not palpable. In the blood at this stage, moderate formation of red blood cells predominates. In the bone marrow, an increase in all sprouts of blood formation. Vascular and visceral complications at this time are possible, but not frequent.
Isolation of the initial (I) stage of erythremia is conditional. In essence, this is a stage with low-symptom manifestations, more characteristic of elderly patients. The spleen is usually not palpable, but its study often reveals a slight increase. Thrombotic complications are possible in this stage of the disease.
IIA stage of the process – erythremic – is unfolded, for it the myeloid transformation of the spleen is uncharacteristic. The duration of this stage is 10-15 years or more. The volume of circulating blood is increased, the spleen is enlarged, and a liver enlargement is somewhat earlier possible. Thrombosis of arterial and venous vessels, hemorrhagic complications at this stage are observed more often. A blood test indicates a “pure” erythrocythemia or erythrocythemia and thrombocytosis or panmyelosis and neutrophilia with stab shift, an increase in the number of basophils. In the bone marrow, total three-growth hyperplasia with severe megakaryocytosis is observed, reticulin and focal collagen myelofibrosis are possible.
The erythremic, developed process, but with myeloid metaplasia of the spleen, also belongs to stage IIB. An increase in blood volume can be expressed to a greater or lesser extent, an increase in the liver and spleen is observed. In the blood at this stage, there is an increase in red blood cells, platelets with leukocytosis above 15 × 103 in 1 μl and a shift in the leukocyte formula to myelocytes, single red blood cells. In the bone marrow, as in stage IIA, an increase in granulocytic germ may predominate, and reticulin and focal collagen myelofibrosis is possible.
In the clinical picture, allergic complications and urate diathesis are often leading.
At this stage, depletion of the patient, exacerbating thrombotic complications and bleeding can be observed.
The III stage of erythremia is called anemic. In the bone marrow, myelofibrosis can be expressed, myelopoiesis is preserved in some cases, and reduced in others. In the enlarged spleen and liver, a myeloid transformation is observed. Erythremia in this stage can result in acute leukemia, chronic myelogenous leukemia, hypoplastic hematopoiesis, and difficultly classified hematological changes.
Arterial hypertension that occurs with erythremia in 35-50% of cases is caused by an increase in peripheral resistance in response to increased blood viscosity, the development of uratic diathesis, chronic pyelonephritis, circulatory disorders in the renal parenchyma, thrombosis and sclerosis of the renal arteries.
Erythremia-specific skin itching associated with washing is observed in 50-55% of patients. In many patients, it becomes the main complaint, it arises not only from contact with water, but also spontaneously, affects performance.
Common complications of the advanced stage of the disease are microcirculatory disorders with a clinic of erythromelalgia, transient disorders of cerebral and coronary circulation and hemorrhagic edema of the legs, as well as venous and arterial thrombosis and bleeding. Already at this stage there may be violations of hemostasis, which often look like latent thrombogenic danger, detected only in the laboratory and without clinical manifestations. At the same time, hemostatic disorders can be even more pronounced, leading to local intravascular coagulation as microthromboses or to disseminated intravascular coagulation – DIC.
The mechanism of development of thrombotic complications of erythremia consists in increasing the mass of circulating red blood cells, slowing down the blood flow and increasing its viscosity. Thrombocytosis and qualitative platelet disturbances contribute to their development. In the blood plasma, circulating aggregates of platelets are often determined, which is the result not only of their quantitative increase, but also of a violation of the functional properties of platelets.
Hemorrhagic complications of erythremia are completely eliminated in patients treated with bloodletting, when the hematocrit index is normalized.
With the development of erythremia, iron deficiency is often observed, eliminating plethora. Clinical manifestations of iron deficiency – weakness, inflammation of the tongue, decreased resistance to infections, thinning nails – are more often observed in older people.
The development of the anemic stage is preceded by a certain dynamics of clinical and hemorrhagic data, in particular, an increase in the spleen, a gradual decrease in plethora, and the appearance of a leukoerythroblastic picture of peripheral blood. In the bone marrow, myelofibrosis gradually develops, which may be accompanied by a type change, cell proliferation, an increase in bone marrow pathology and hemopoiesis inefficiency – the outcome of erythremia in secondary myelofibrosis.
There are other forms and variants of the course of the disease, in which from the very beginning an increase in the spleen due to myeloid transformation is detected. Exacerbations of the disease after treatment with cytostatics proceed mainly with plethora and an enlargement of the spleen. These are always pancytic forms of the disease with leukoerythroblastic blood picture, more severe than ordinary erythremia.
They differ from erythremia by early and pronounced extra-bone marrow distribution, a greater three-growth focus of growth and reticulin myelofibrosis, and from idiopathic myelofibrosis by the presence of plethora and duration of myeloproliferation, by the absence of a tendency to the rapid completion of reticulin myelofibrosis.
However, anemia that develops with erythremia may have a different development mechanism, is not always associated with the progression of the process, and in many cases it is successfully treated.
Anemia can be iron deficiency due to bleeding and bloodletting; hemodilution, associated with an increase in the volume of circulating plasma due to an increase in the spleen, hemolytic, caused by increased function of the spleen. Finally, anemia with erythremia may be the result of ineffective hematopoiesis. With the outcome of erythremia in acute leukemia or in hemopoiesis hypoplasia, anemia characteristic of these processes is observed.
The frequency of the outcome of erythremia in acute leukemia is 1% in untreated and 11-15% in those treated with cytostatics (chlorobutin), more often acute myeloid leukemia and erythromyelosis develop. Harbingers of acute leukemia, sometimes occurring 2-3 years before its diagnosis, are non-infectious fever, unmotivated leukopenia, thrombotic or pancytopenia, sometimes dermatitis.
Post-erythremic myelofibrosis is the result of the natural evolution of the disease. It is observed in every patient with erythremia surviving to this period. The difference in its hematological manifestations and course is striking – from benign, with hematological compensation, to malignant, with rapid anemization, depression of granulo- and thrombocytopoiesis, sometimes with low-percentage blastemia. In these cases, it is likely that tumor progression of the disease should be assumed, for months and years to go through manifestations of which in the form of a blast crisis.
Diagnosis of Erythremia
Diagnosis of erythremia is complicated by the fact that it is not the only cause of erythrocytosis.
The following types of anemia are distinguished.
- Erythremia.
- Secondary absolute erythrocytoses (due to increased formation of erythropoietins).
- With generalized tissue hypoxia (hypoxic, compensatory):
– with arterial hypoxemia: “high-altitude” disease, chronic obstructive pulmonary disease, congenital “blue” heart defects, arteriovenous anastomoses, carboxyhemoglobinemia (mainly due to tobacco smoking);
– without arterial hypoxemia: hemoglobinopathies with increased affinity for oxygen, deficiency of 2, 3-diphosphoglycerate in red blood cells.
For tumors: kidney cancer, cerebellar hemangioblastoma, Hippel-Lindau syndrome, hepatoma, uterine fibroids, tumors of the cortical and brain layers of the adrenal gland, pituitary adenoma and cyst, masculinizing ovarian tumors.
With local renal ischemia (dysregulatory): kidney cysts (solitary and multiple), hydronephrosis, renal transplant rejection, renal artery stenosis.
- Cobalt (mostly experimental).
- Secondary relative, hemoconcentration erythrocytosis: stress erythrocytosis, Geysbek syndrome, pseudopolycythemia.
- Primary erythrocytosis.
Erythremia is diagnosed by certain standardized criteria. Erythremia can be suspected by an increase in red blood and hematocrit in the peripheral blood: for men more than 5.7 × 106 red blood cells in 1 μl, HB more than 177 g / l, Ht 52%; for women more than 5.2 × 106 red blood cells in 1 μl.
The criteria for the diagnosis of erythremia are as follows.
- An increase in the mass of circulating red blood cells: for men – more than 36 ml / kg, for women – more than 32 ml / kg.
- Normal saturation of arterial blood with oxygen (more than 92%).
- Enlarged spleen.
- Leukocytosis more than 12 × 103 in 1 μl (in the absence of infections and intoxications).
- Thrombocytosis more than 4 × 105 in 1 μl (in the absence of bleeding).
- An increase in the content of alkaline phosphatase of neutrophils (in the absence of infections and intoxications).
- Increase in unsaturated vitamin B 12-binding ability of blood serum.
The diagnosis is reliable with 3 any positive signs.
With plethora, enlargement of the spleen, leukocytosis and thrombocytosis, the diagnosis of erythremia is not difficult, however, even in these cases, an examination of the ilium is necessary to confirm the diagnosis and comparative diagnosis with other myeloproliferative diseases.
Diagnostic problems arise in relation to purely erythrocytemic forms of polycythemia without enlarged spleen, which can turn out to be both erythremia and erythrocytosis: about 30% of patients with erythremia do not have leukocytosis and thrombocytosis in the diagnosis.
For comparative diagnosis, a radiological measurement of the mass of circulating red blood cells, and sometimes the volume of circulating plasma using serum albumin is necessary.
When a normal mass of circulating red blood cells and a reduced plasma volume are detected, a relative increase in red blood cells is diagnosed.
Relative erythrocytosis should be assumed when, with elevated levels of red blood, patients have the usual color of the skin and mucous membranes.
With an increase in the mass of circulating red blood cells, a comparative diagnosis is made between erythremia and absolute red blood cells. In smokers, a study of the content of carboxyhemoglobin is carried out in the morning, afternoon and evening, as well as 5 days after smoking cessation.
With the exclusion of hypoxic erythrocytosis, the object of study should be the kidneys, and then other organs and systems whose diseases are accompanied by erythrocytosis.
A histological examination of the ilium allows the doctor to establish the correct diagnosis in 90% of cases. Occasionally, there is no change in bone marrow with erythremia, and then the doctor can diagnose erythremia only with a convincing clinical and hematological picture.
For a comparative diagnosis of erythremia and erythrocytosis, erythropoietins are examined, the amount of which is reduced with erythremia, and increased with erythrocytosis.
The morphological and functional characteristics of blood cells should be considered. Erythremia is confirmed by large forms of platelets and a violation of their aggregation properties; an increase in the number of neutrophils over 7 × 103 in 1 μl; increase in the content of alkaline phosphatase in them; detection of a high content on the membrane of neutrophils of receptors for IgG; increased lysozyme content; an increase in the absolute number of basophils (staining with acrylic blue) over 65 / μl; an increase in the content of gluetamine in the blood and urine (a product of basophil secretion).
Patients for whom the causes of polycythemia could not be determined should belong to the group of patients with unclassified polycythemia. Cytostatic treatment is not indicated for such patients.
Erythremia Treatment
The goal of the treatment is to normalize the amount of hemoglobin to 140-150 g / l (85-90 PIECES) and the hematocrit index (46-47%), since it is precisely in this case that the risk of vascular complications decreases sharply. Bloodletting is prescribed 500 ml every other day in a hospital and after 2 days with outpatient treatment. Instead of bloodletting, it is better to carry out erythrocytapheresis. The amount of bloodletting is determined by the achievement of normal red blood counts.
In elderly patients, or with concomitant diseases of the cardiovascular system, or poorly tolerating bloodletting, no more than 350 ml of blood is removed once, and the intervals between bloodletting are somewhat lengthened. To facilitate bloodletting and the prevention of thrombotic complications on the eve and on the day of the procedure or during the entire period of bloodletting, as well as 1-2 weeks after the end of treatment, disaggregant therapy should be prescribed – acetylsalicylic acid 0.5-1 g / day and chimes 150-200-200 mg / day at the same time. In addition, immediately before bloodletting, the introduction of 400 ml of reopoliglyukin is recommended.
With contraindications to the use of acetylsalicylic acid, the doctor prescribes chimes, papaverine or nicotinic acid preparations. At the end of treatment, the condition of the patients and the blood picture are monitored every 6-8 weeks.
Indications for the appointment of cytostatics are erythremia with leukocytosis, thrombocytosis and an enlarged spleen, skin itching, visceral and vascular complications, the patient’s serious condition, as well as the insufficient effectiveness of the previous treatment for bloodletting, the need for their frequent repetition, poor tolerance and complication of stable thrombocytosis, clinically manifest iron deficiency. In the latter case, against the background of treatment with cytostatics, iron replacement therapy is carried out. The elderly patients (over 50 years), the inability to organize bloodletting therapy expand the indications for treatment with cytostatics.
Cytostatic therapy is usually combined with bloodletting, prescribed before the normalization of hematocrit and the amount of hemoglobin from the very beginning of cytostatic therapy.
Hematological monitoring of the course of treatment is carried out weekly, and by the end of treatment – every 5 days.
Maintenance therapy with cytostatics is not recommended due to the low effectiveness and the risk of leukemic effects. Preferred timely course treatment in full or reduced volume with a tendency to exacerbations.
Uratic diathesis is an indication for the appointment of milurite (allopurinol) in a daily dose of 0.3 to 1 g. The drug reduces the synthesis of uric acid from hypoxanthine, the content of which increases due to cellular hypercatabolism. In the treatment of cytostatics, the drug is prescribed prophylactically in a daily dose of 200 to 500 mg or more.
Microcirculatory disorders and, in particular, erythromelalgia (attacks of sudden burning pain mainly in the extremities with local redness and swelling of the skin), caused mainly by the aggregation block of arterial blood flow in the capillaries and small arteries, are successfully treated with acetylsalicylic acid at 0.31 g per day. The effectiveness of chimes alone with erythromelalgia is significantly lower.
It should be noted that gastrointestinal bleeding has appeared in connection with the widespread use of acetylsalicylic acid, including prolonged and posing a real danger. Long nose and gingival bleeding is possible.
This complication of treatment is caused by both unrecognized ulcerative lesions of the gastrointestinal tract, characteristic of erythremia and asymptomatic, and the initial functional defect of platelets aggravated by acetylsalicylic acid.
Acute vascular thrombosis is an indication for the appointment of not only platelet disaggregants, but also heparin, transfusions of freshly frozen plasma.
When treating in the anemic phase, the mechanism of the development of anemia, thrombocytopenia and other symptoms is taken into account. In case of anemia caused by iron or folic acid deficiency, appropriate replacement therapy is prescribed. Treatment of hemodilutionary anemia should be aimed at reducing the spleen using radiation therapy, cytostatics, and prednisone. Anemia caused by insufficient red blood cell formation is preferably treated with androgens or anabolic steroids. Prednisolone is prescribed mainly for suspected autoimmune origin of anemia and thrombocytopenia, as well as to reduce the spleen.
2 treatment regimens are used:
- the appointment of a high dose of prednisolone – 90-120 mg / day for 2 weeks, followed by a transition to medium and small doses with the effect and drug withdrawal in case of inefficiency;
- the appointment from the very beginning of the average daily doses (20-30 mg), and then small doses (15-10 mg) for 2-3 months with the mandatory withdrawal of the drug. In many cases, a clear positive effect of steroid therapy is observed, although the mechanism of its action is not completely clear.
For outcomes in acute leukemia, polychemotherapy is used taking into account the histochemical variant, and for outcomes in a typical and atypical myeloid leukemia, myelosan and myelobromol, hydroxyurea, but with little effect. In post-erythremic myelofibrosis, increasing leukocytosis and the progression of splenomegaly, short courses of therapy with myelobromol (250 mg / day) or myelosan (4-2 mg / day for 2-3 weeks) are advisable.
With anemic and thrombocytopenic syndromes, glucocorticosteroids are used, often in combination with cytostatics (in small doses) with suspected enlargement of the spleen. For the same purpose, γ-therapy can be applied to the spleen area at a course dose of 5 Gy, sometimes slightly more if the number of platelets allows. A positive effect of small doses of prednisone (15-20 mg / day), prescribed for 2-3 months, on the size of the spleen, general manifestations of the disease and blood picture, was noted, but it is limited to the treatment period and the shortest time after its withdrawal.